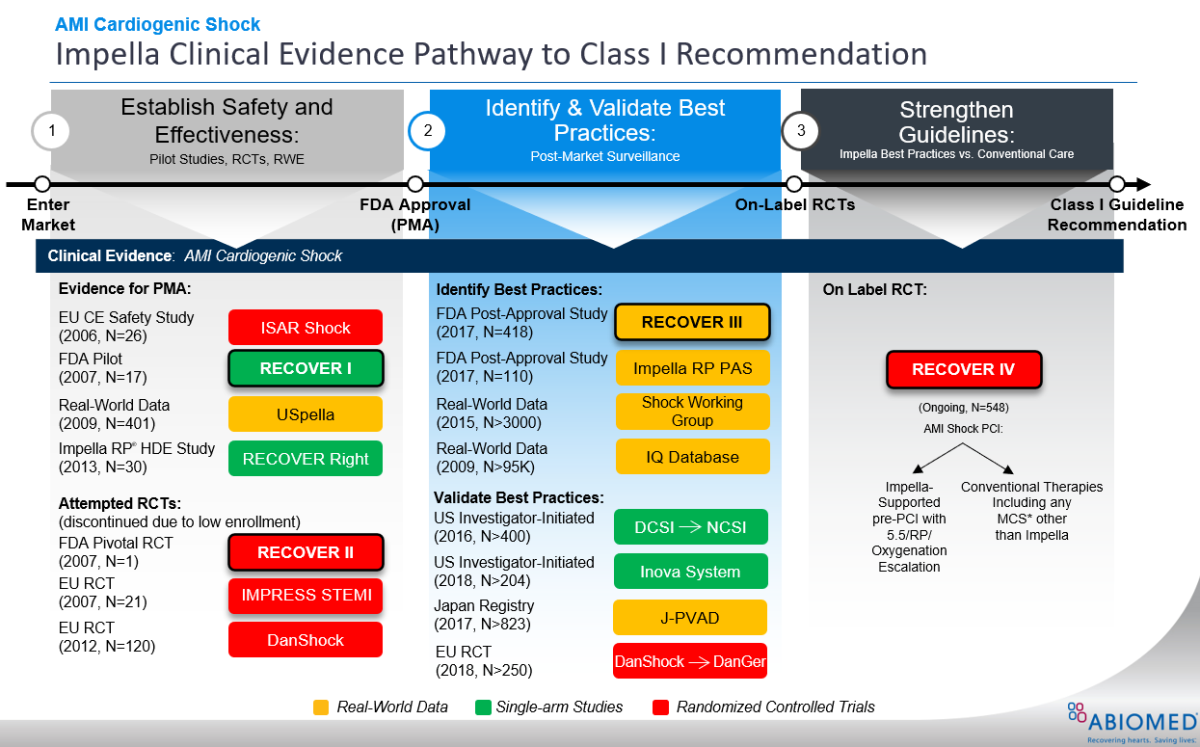
Metavention's Renal Denervation Pivotal IDE Study now Approved by FDA for the Treatment of Hypertension
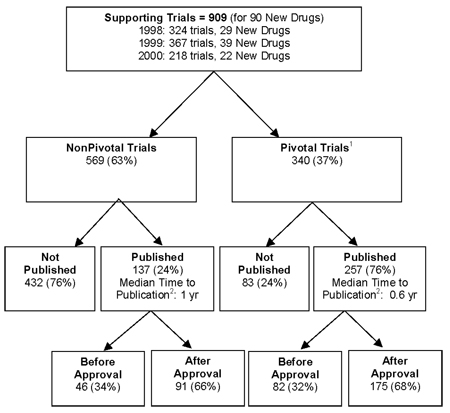
Registration, publication, and outcome reporting among pivotal clinical trials that supported FDA approval of high-risk cardiovascular devices before and after FDAAA | Trials | Full Text

FDA Approval of IDE to Initiate Multi-Center Pivotal Study of Alpha DaRT™ in Recurrent Cutaneous SCC

Early feasibility studies in the United States: Focus on electrophysiology - Weiss - 2023 - Journal of Cardiovascular Electrophysiology - Wiley Online Library

Mainz Biomed Announce IRB Approval and Initiation of US Pivotal FDA Clinical Study | Technology Networks
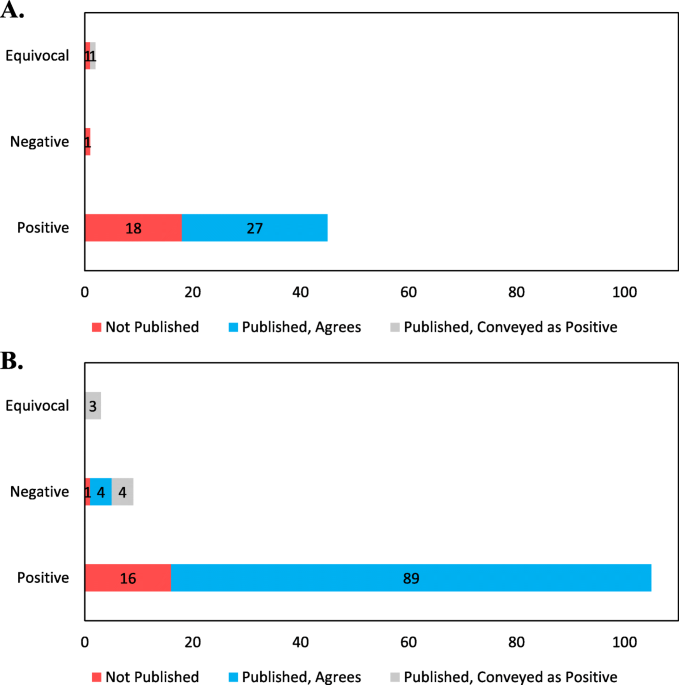
Registration, publication, and outcome reporting among pivotal clinical trials that supported FDA approval of high-risk cardiovascular devices before and after FDAAA | Trials | Full Text

Early phase clinical trial played a critical role in the Food and Drug Administration–approved indications for targeted anticancer drugs: a cross-sectional study from 2012 to 2021 - ScienceDirect

PQ Bypass Announces Unconditional IDE Approval from U.S. FDA for Pivotal Study. - Seroba Life Sciences

FDA Guidance on Design Considerations for Pivotal Clinical Investigations: Non-Comparative Clinical Outcome Studies | RegDesk

Early Feasibility Studies for Cardiovascular Devices in the United States: JACC State-of-the-Art Review - ScienceDirect

Sample construction for pivotal trials using surrogate markers with... | Download Scientific Diagram

The Winding Journey From EFS/FIH to Pivotal Study: How to Work Closely With the FDA Through the Ups and Downs | tctmd.com

Early Feasibility Studies for Cardiovascular Devices in the United States: JACC State-of-the-Art Review - ScienceDirect